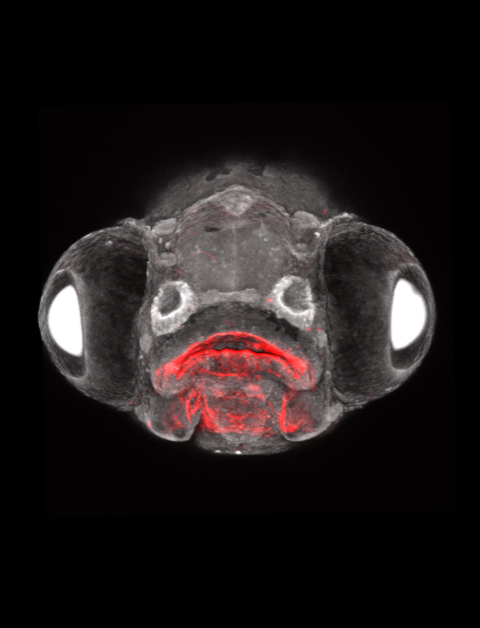
My lab, based at the University of Southern California Keck School of Medicine, uses zebrafish to model human birth defects affecting the face. When I tell people this, they are often skeptical that fish biology has any relevance to human health.
But zebrafish have backbones like us, contain by and large the same types of organs, and, critically for genetic research, share many genes in common. My group has exploited these genetic similarities to create zebrafish models for several human birth defects, including Saethre-Chotzen Syndrome, in which the bones of the skull abnormally fuse together, and early-onset arthritis.
Similar to fish, our bodies develop under the control of about 25,000 genes. The trick is finding out what each gene does. Stunning advances such as CRISPR-based molecular scissors, for which the Nobel Prize in chemistry was just awarded, allow us to precisely change genes, and designer chemicals can silence particular genes. In a recent study from our group published in Nature, however, we find that these tools are still far from perfect. Although CRISPR now allows us to efficiently generate lab animals that can pass human disease mutations onto the next generation, claims that simply injecting CRISPR into embryos or silencing genes with designer chemicals can accurately model human genetic disease are being questioned.

Emergence of zebrafish as a model for human genetic disease
Finding the precise mutation that causes a particular birth defect or a late-onset disease can be tedious work. The human genome is made up of 3 billion building blocks called DNA nucleotides, and changing just one of these can cause devastating birth defects.
To figure out if we have identified the right disease-causing mutation in humans, we typically engineer the same change into the genome of a lab animal. We then breed these animals to generate babies with the disease mutation and look for the appearance of defects similar to those in human patients.
We study zebrafish because they are small, which means we can grow thousands of different genetically modified animals. We routinely use CRISPR to engineer fish that pass on a gene-breaking mutation to the next generation.
We then study the appearance of defects similar to those in humans lacking these genes – in essence creating personalized zebrafish avatars of genetic disease. As zebrafish embryos are transparent and develop rapidly outside the mother, they are particularly useful for understanding how human disease mutations disrupt normal development.

In the race for speed, problems emerge
Even in zebrafish, engineering animals to lack particular genes can be a time-consuming process. In my lab, we first create gene mutations in embryos, grow these fish to adulthood and then breed fish together to look at defects in the next generation.
This whole process can take a year or longer. Unsurprisingly, many labs are attempting shortcuts. Some are injecting large quantities of CRISPR molecular scissors into animals and then looking for defects in these same animals. Others are using chemicals to turn off, or silence, genes in the embryo rather than permanently changing the genes.
More and more frequently studies like this are calling into question the accuracy of these shortcuts. In animals that have been injected with CRISPR molecular scissors, not every cell is changed in the same way. And the chemicals used to silence genes appear to have unintended consequences, poisoning the embryo in a generic way.
For example, researchers in Spain recently reported that a gene called prrx1a was critical for the proper development of the heart. To figure this out, they silenced prrx1a in zebrafish with chemicals. Then, in a second experiment, they injected CRISPR molecular scissors into zebrafish embryos and examined them just one day later for heart defects.
In contrast, we completely removed the prrx1a gene and looked at generations of fish lacking this gene. Hearts in these mutant fish developed perfectly normally, showing that prrx1a was not critical for heart development. Instead, we showed that the heart defects seen upon chemical treatment in the Spanish study were due to a general poisoning of the embryos unrelated to the prrx1a gene. Animals simply injected with CRISPR also showed defects not seen upon complete removal of the prrx1a gene, although the exact reasons for these differences remain a source of active debate.
And not just our group has noticed these flaws. Using similar gene removal as we reported, the group led by Didier Stainier refuted a study that had used CRISPR injection and gene silencing to link the tek gene to blood vessel development. Given the number of studies relying on gene silencing in lab animals, as opposed to engineering the DNA mutations, the causative genes for many human diseases may need to be reevaluated.
A path forward with improved genome engineering
The desire for speed in research must not come at a cost of accuracy and reproducibility.
The good news is that, with the ease of CRISPR, we now know how to engineer the right types of mutations in lab animals to validate human disease mutations. By creating lab animals such as zebrafish that have the mutations engineered into their genomes and then observing whether their offspring develop the same diseases as patients with the mutations, we can be confident in having identified the right human disease gene.
Getting it right is important for accurately counseling prospective parents of their genetic risks for certain birth defects, as well as identifying the relevant genes that can be targeted to prevent or even reverse disease.
Science is constantly evolving. While the ability to engineer the genome with CRISPR is opening up endless possibilities for human genetics, researchers must also recognize the limitations of new technologies. Although rapid, directly injecting CRISPR or silencing genes with chemicals gives misleading results too often. In order to confidently identify causative mutations linked to human disease, we will need to continue to study lab animals engineered to carry and pass on the same DNA changes as found in human patients.