Anyone with arthritis can appreciate how useful it would be if scientists could grow cartilage in the lab. To this end, Keck School of Medicine of USC scientists in the USC Stem Cell laboratory of Denis Evseenko, MD, PhD, collaborated with colleagues at several institutions to provide new insights into how gene activity drives the development of cartilage. Their findings appear today in Nature Communications.
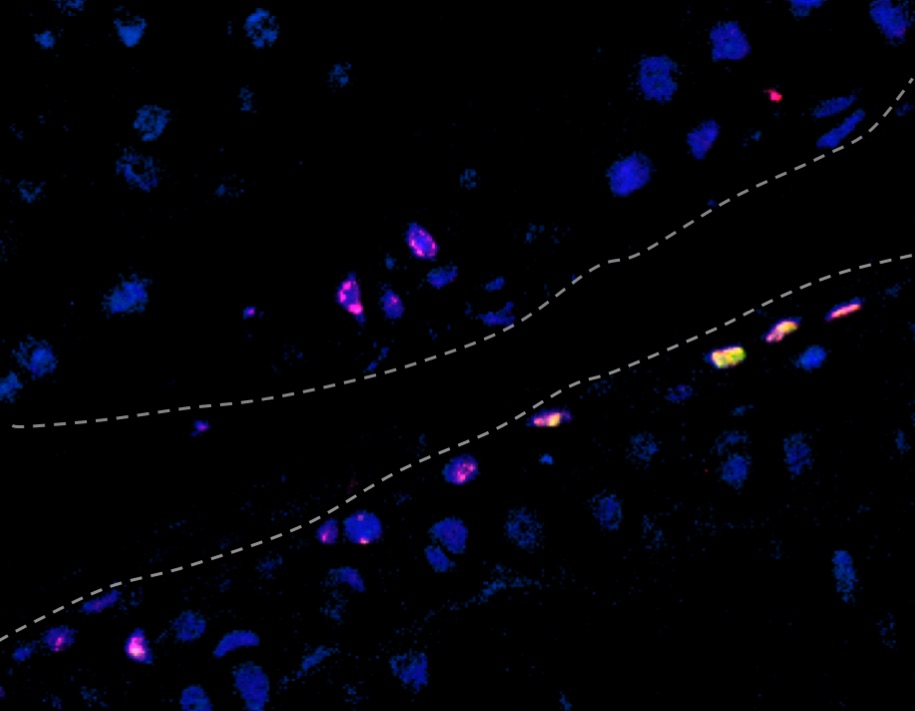
Based on their molecular studies, the scientists identified and characterized, for the first time, unique cell populations that form the superficial zone of human joint cartilage. The superficial zone plays the most critical role in cushioning the joint, and is often partially or completely lost in arthritis.
“Our results not only offer a unique molecular atlas of human skeletal development, but also define a strategy for joint cartilage repair,” says Evseenko, who is the study’s corresponding author and an associate professor of orthopaedic surgery, and stem cell biology and regenerative medicine at the Keck School.
In a series of experiments, Gabriel B. Ferguson, PhD, postdoctoral scholar – research associate, Ben Van Handel, PhD, postdoctoral scholar – research associate, and colleagues compared the gene activity of developing human cartilage cells with several other cell types.
First, they compared the cartilage cells to four other types of developing human cells: the precursors to bone, muscle, tendon and ligament. As the cartilage matured, the genes specific to cartilage became increasingly active, while genes related to the other cell types became repressed.
Next, the scientists compared these developing human cartilage cells to equivalent cells from mice. The team found many broad similarities in gene activity.
The researchers also carried out a detailed comparison of ordinary human cartilage cells and stem cell–derived human cartilage cells, taking into account genetics, genetic regulation and function. The researchers demonstrated that stem cell–derived cartilage does not fully develop in the Petri dish but rather retains the genetic hallmarks typical of fetal cartilage. However, if the scientists transplanted stem cell–derived human cartilage at a particular stage of development into an arthritic rat, the cartilage would lose its fetal hallmarks and fully mature — regenerating the critical superficial zone.
Additional authors include: Maxwell Bay, Siyoung Lee, Ruzanna Shkhyan, Nicholas W. Banks, Mila Scheinberg, Biagio Saitta, Joseph Elphingstone and Michael Bonaguidi from USC; Petko Fiziev, April Pyle, Nicholas M. Bernthal, Hanna KA Mikkola and Jason Ernst from UCLA; Tonis Org from UCLA and the University of Tartu in Estonia; Ling Wu from InVitro Cell Research; and A. Noelle Larson, Scott M. Riester and Andre J. van Wijnen from the Mayo Clinic.
This study was supported by the National Institutes of Health under award number R01AR071734, the U.S. Department of Defense under award number W81XWH-13-1-0465 and the California Institute for Regenerative Medicine (CIRM) under award numbers RB5-07230-B and TRAN1-09288. Approximately 50 percent of the project’s funding was federally funded. Fifty percent of the project’s funding was not federally funded. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.