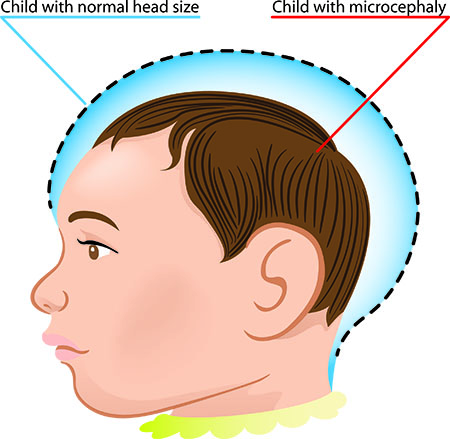
Microcephaly is a condition where the circumference of an individual’s head is smaller than normal. It can be caused by genetic abnormalities as well as fetal exposure to drugs; alcohol; certain viruses, such as Zika virus; or toxins during pregnancy.
It’s rare — there are fewer than 200,000 cases a year in the United States. Assistant Professor Jianfu “Jeff” Chen aims to better understand the mechanisms that cause microcephaly — in particular, the ways in which specific genetic mutations create this condition.
Chen has been interested for the past few years in better understanding how the disorder develops. “Often times, patients with microcephaly also have intellectual disability, but the cause of the smaller brain size remains to be established,” he said.
In 2014, Chen created a mouse model for microcephaly — but found that the difference between a microcephalic mouse brain and a regular mouse brain wasn’t a large one. “Using the mouse model, we thought we were missing some component that was specific to human brains,” he said.
Tiny, artificial brains
In a new paper published in Nature Communications, Chen adopted a new model system: a brain organoid. Brain organoids are artificially-grown miniature organs that resemble the brain, created by culturing human pluripotent stem cells in a three-dimensional bioreactor and developed over months. “We can create three-dimensional brain tissue to model early human brain development,” Chen explained.
Using this system, he was able to tease out the dramatic size difference in a brain when a specific gene — WDR62 — is knocked out. “Basically, we discovered a new mechanism to explain why microcephaly develops,” he said. The mutation of this gene changes many things — including how many tiny hairs, called cilia, are produced in the brain cells — and how long they are.
“Because you have too many cilia and they stay longer than they’re supposed to, the cell cycle is delayed,” Chen explained. This delay causes less cell division and leads to the production of fewer neurons, which contributes to a smaller brain size in patients.
Why is the disorder often accompanied with intellectual disability?
Chen’s next step is to understand why microcephaly is often accompanied with intellectual disability. Most children born with microcephaly have intellectual disability, but not all. “We are trying to understand how that happens: why the mutation of certain genes leads to behavior disorder,” Chen said. He hopes to connect the particular quirk of cilia dysregulation to intellectual disability.
His ultimate goal is to understand the cause of this microcephaly disease and identify potential set of treatments. One possibility for the future is to focus strategies on a downstream mediator, which can reduce cilia length and numbers.
Using the brain organoid lets the researchers understand brain development on a whole new level — showing the process in far more detail than mouse models. “This work is important because, for the first time, we can model this WDR62-related human brain disorder in a culture dish and discover some novel disease causes that cannot be easily identified in mouse models,” Chen said.
