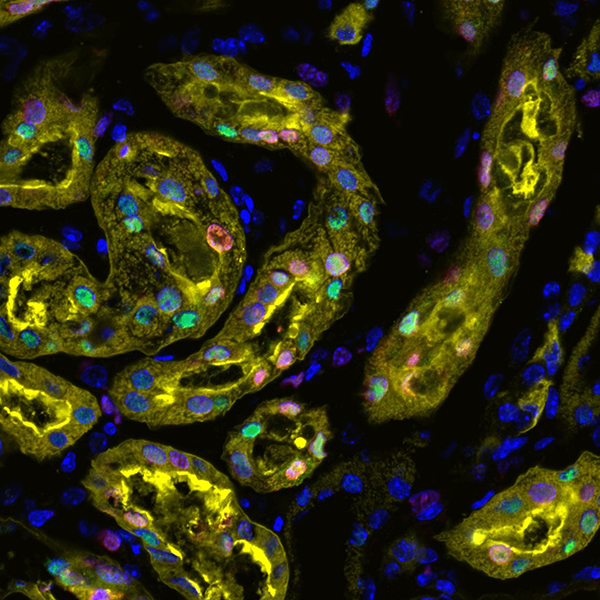
In the kidney, injured cells can be kicked into reparative mode by a gene called Sox9, according to a new paper published in Cell Reports.
First author Sanjeev Kumar, a postdoctoral research associate in the USC Stem Cell laboratory of Andy McMahon, found that surviving injured cells switch on the Sox9 gene as a response to kidney damage. This regenerates the injured cellular lining of the nephron, the functional unit of the kidney, and repairs the kidney after acute kidney injury (AKI).
By recruiting the majority of the surviving cells of the epithelium to aid in the timely repair of a severely injured organ, the kidney’s Sox9 strategy contrasts with the stem cell-based repair strategy of many other organ systems.
“Currently, no treatment exists to treat AKI per se. Identifying the kidney’s intrinsic mechanisms of repair is critical for developing treatments to kickstart the kidneys after AKI, a serious condition with an in-hospital mortality rate exceeding 50 percent,” said Kumar.
In sections of the kidney that fail to repair, Sox9 remains activated thereby, demarcating regions of inefficient repair responses. Further interrogation of such regions could provide a crucial link between AKI and its transition to chronic and end-stage kidney disease.
Sox9 also plays a key role in the normal development of the kidney.
Additional co-authors include Jing Liu, Paul Pang, A. Michaela Krautzberger and Andy McMahon from USC; Antoine Reginensi and Andreas Schedl from the Centre de Biochimie in Nice, France; Haruhiko Akiyama from Gifu University in Gifu, Japan; and Benjamin D. Humphreys from Harvard Medical School.
Kumar is the recipient of a John McKay Fellowship Award from the University Kidney Research Organization, Los Angeles. Schedl received grants from the Agence nationale de la recherche (09-Geno-027-01; ANR-11-LABX-0028-01), and both Schedl and Reginensi received support from the Fondation pour la recherche médicale. The McMahon laboratory received funding from the California Institute for Regenerative Medicine (LA1-06536).